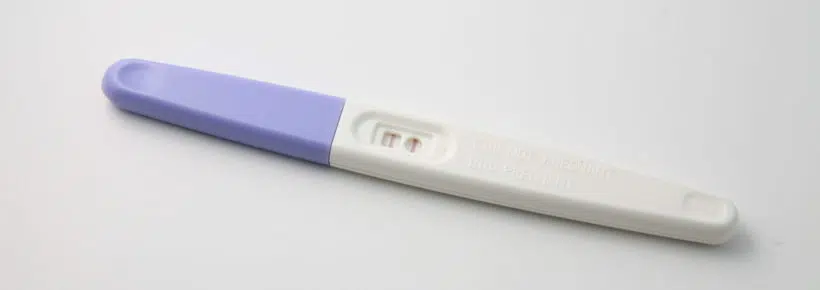
The intrauterine device (IUD) is a foreign body inserted into the uterus. IUDs are made of a non-reactive plastic such as polyethylene and may include active chemicals or elements such as progesterone or copper which slowly diffuse into the uterus over a period of several years.
Human Life International has a free e-book, How Contraception Built the Abortion Culture, that goes into detail on how IUDs contribute to the abortion rate. You can download it now for free to learn more about the abortion-contraception link.
Is the IUD Abortifacient?
Short answer: sometimes.
The intrauterine device has several contraceptive (non-abortive) effects:
- It prevents sperm from fertilizing ova
- It releases ions that interfere with fertilization
- It thickens the cervical mucus
- It inhibits sperm capabilities
These are all purely contraceptive effects.
But IUDs also irritate the endometrium (the lining of the uterus) and make it inhospitable to the blastocyst (the very early developing human being), which is an abortifacient effect.1

Nearly half a century ago, the American Medical Association recognized that “the action of the IUDs would seem to be a simple local phenomenon. That these devices prevent nidation [implantation] of an already fertilized ovum has been accepted as the most likely mechanism of action.”2 The Food and Drug Administration (FDA) concurred in this assessment, finding that “IUDs seem to interfere in some manner with the implantation of the fertilized egg in the lining of the uterine cavity. The IUD does not prevent ovulation.”3
Nothing has changed. The patient information pamphlets for all of the IUDs made today describe in detail this abortifacient action. For example, the PIP for the Skyla IUD says that one of its mechanisms of action is “alteration of the endometrium.”
In summary, every IUD ever manufactured prevents implantation, meaning that they are all abortifacients.
These devices ensure that the uterus is as barren and hostile to new life as a desert. In fact, this abortifacient action is so efficient that scientists recommending the IUD for “emergency contraception” note that only one in 1,100 women will remain pregnant after inserting the device from two days to two weeks after their last unprotected intercourse.4
Controversy over the IUD
The first modern-day IUD, the Grafenberg Ring, was placed on the market in 1915. Several other companies immediately saw the opportunity for profit, and by the mid-1930s, there was a proliferation of IUDs for sale. They sported intimidating names such as the Sterilette, the Collar-Button, the Wishbone, the Silkworm Pust, and the Star.
Some of the devices were of good quality, but many were made of “cheaper, noncorrosive metals, or of celluloid, bakelite, and chromium-plated metals rather than gold.”5
According to one expert at the time, “Sometimes these [cheaper IUDs] were claimed to be made of solid gold in order to enhance the price, and a few practitioners took economic advantage of the need and ignorance of women.”6
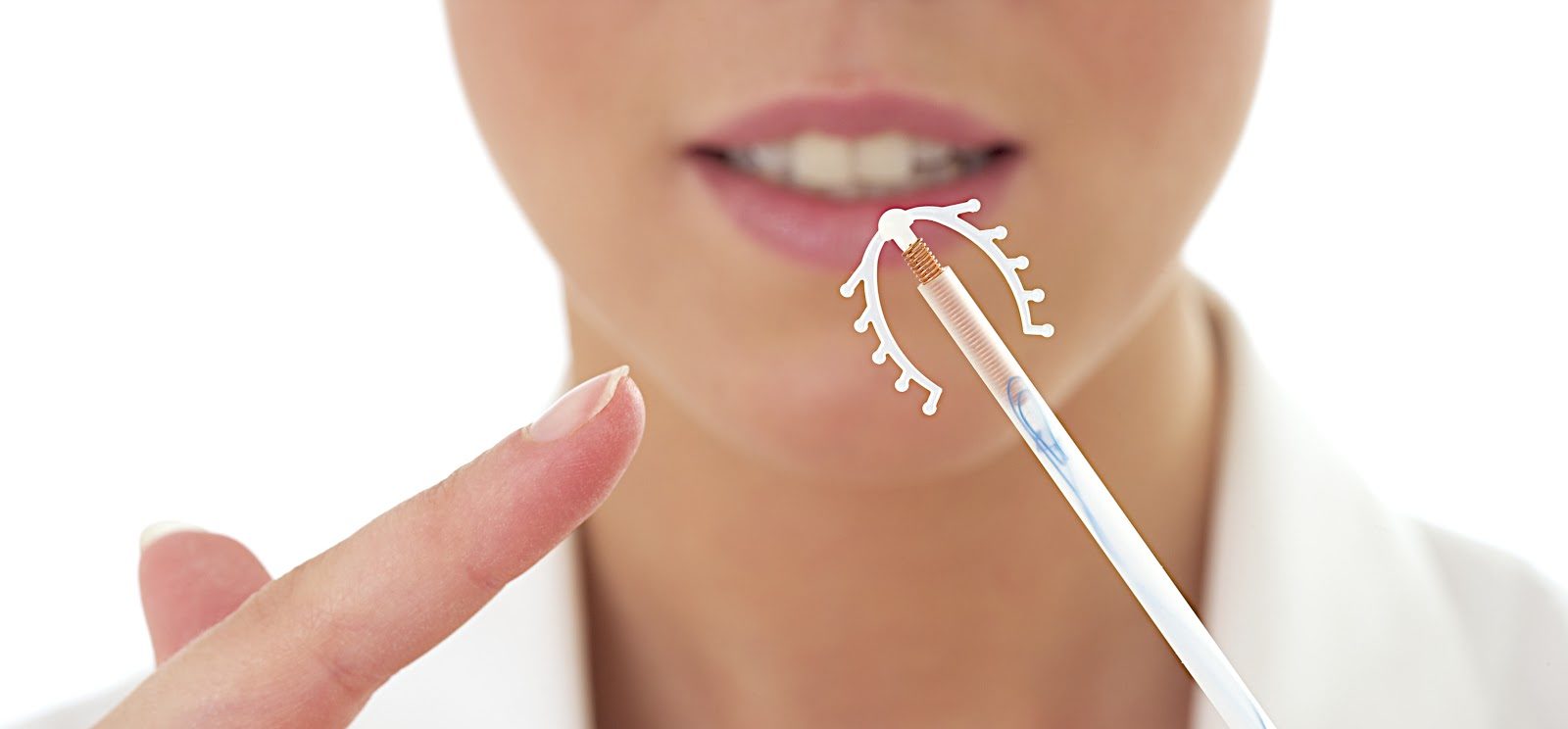
More than 70 different types of IUDs have been manufactured over the last century. Some of these have consisted of polyethylene with barium sulfate so they could be detected by X-rays. The Dalkon Shield, which was the most popular IUD in use for several years in the United States, was of this type. It was withdrawn from the market in 1984 due to a number of maternal deaths.7
Other IUDs, including the Progesterone-T, were loaded with varying doses of progesterone crystals that were usually suspended in silicone oil. This IUD released about 24 milligrams of progesterone a year. It was originally promoted under the truly Orwellian label “Uterine Therapeutic System.”8
The copper IUDs (including the “Copper-7,” “Copper-T” and “Tatum-T”) discharged from 50 to 75 micrograms of ionic copper into the uterus each day. These copper ions interfere with the life-sustaining functions that regulate implantation of the new human life in the uterus. Copper has been proven to be the active agent in these IUDs because identical devices are ineffective without the element. Each copper IUD is effective at causing early abortions for about four years.9
There are five IUDs currently [in 2022] available on the American market:
- The first type is the non-hormonal Copper T 380A (ParaGard), which consists of a flexible T-shape containing copper collars on both arms and a copper coil along the stem and is supposedly good for up to ten years of sterility.
- The second type of IUD is loaded with a total of 52 milligrams of levenorgestrel (LNG an artificial progestin) and releases it at a rate of about 20 micrograms per day, declining to 14 micrograms per day after five years.10 Liletta and Mirena are in this class of IUD.
- The third type is the lower-dose IUDs, which have 13.5 to 19.5 milligrams of levenorgestrel and are effective for about three years. The Skyla and Kyleena IUDs are in this class.
Complications Associated with IUD Use
According to their patient information pamphlets, complications associated with the IUDs in current use include:
- sterility
- hemorrhage
- perforation of the uterus, colon, bladder, or small or large intestine
- cervical lacerations
- cervical dysplasia (developmental abnormalities)
- deep embedding of the IUD (a serious problem in developing countries, where women may have had the devices in their uteri for a decade or more)
- fragmentation of the IUD
- development of hydatidiform moles
- salpingitis (inflammation of the uterine tubes)
- pelvic inflammatory disease (PID, which often leads to infertility or sterility)
This is nothing new. Until about 1960, the medical community generally condemned IUDs because of their severe side effects. Alan Guttmacher, former President of the Planned Parenthood Federation of America, said in 1959, “Intrauterine devices are mentioned only to be thoroughly condemned because of their ineffectiveness, their potential source for infection and irritation, as well as their carcinogenic potential.”11

Although IUD technology had not advanced in the slightest, the entire medical community performed an abrupt about-face in the mid-1960s and began to accept the device. But serious complications were documented almost immediately. In 1974, the FDA summarized complaints received about the various IUDs on the market at that time.
The FDA had received reports of 238 cases of spontaneous septic abortion from women who had become pregnant with IUDs in place. Of these women, 21 died. The Dalkon Shield was involved in 14 of these deaths and 209 cases of septic abortion, and the Lippes Loop caused 5 deaths and 21 septic abortions.12
Overall, there were about 15,000 IUD-related hospitalizations annually in the early 1970s.13
A.H. Robin Pharmaceuticals made the Dalkon Shield IUD from 1971 to 1974. Documented reports of severe injuries began to surface almost immediately after initial distribution, and on 29 June 1975, the FDA announced it intended to “require special warning notices for users of the intrauterine devices, the contraceptives that were linked to 43 deaths in recent years.”14
A.H. Robin pulled the Dalkon Shield from the market in 1975. By 1985, 13,000 women had sued the company for damages relating to sterility, miscarriages and pelvic infections. Incredibly, some population controllers, including Stephen Mumford and Elton Kessel ― who peddle the dangerous sterilizing chemical quinacrine in developing countries ― have tried to sanitize the image of the Dalkon Shield so that it can make a comeback.15
Ortho Pharmaceuticals withdrew its Lippes Loop from the American market in 1985, and G.D. Searle withdrew its Copper-7 and Tatum-T IUDs in 1986 when its liability insurance lapsed. Searle also faced 775 lawsuits from women who suffered injuries from their IUDs or who conceived babies with birth defects when the devices did not work properly.
Because of widespread negative publicity, only 1% of women using birth control were on the IUD by 1995, but this number had risen to about 9% by 2017.16
Why Is This Abortifacient Called a Contraceptive?
When discussing abortifacient means of birth control, the “family planners” call us pro-lifers “anti-science” when we claim that the IUD causes early abortions. They say that human life begins at implantation, not fertilization.
But there is no logical reason to make this claim. What is the difference between the early human being (blastocyst) the hour before it implants and the hour after?
Whenever there is such confusion, we may find the answer in the dishonest manipulation of terminology by the “family planners.”
In 1963, the United States Department of Health, Education and Welfare (HEW) defined “abortion” as “all the measures which impair the viability of the zygote at any time between the instant of fertilization and the completion of labor.”17
Until the mid-1960s, scientists universally acknowledged that conception happened at the moment of fertilization of the ovum by the spermatozoa somewhere in the Fallopian tube. But pro-abortionists and population controllers already had their sights set on a shift from contraceptive to abortifacient methods of birth prevention, and abortifacient research was already ongoing in Japan and several European countries.
To make abortifacients acceptable to women, and to circumvent laws designed to prohibit abortion, the “family planners” realized that they had to blur the line between contraceptive and abortifacient action.
They could do this only by changing the definition of “conception” from fertilization [union of spermatozoa and ovum] to implantation. Under the new definition of “conception,” if a device or drug ― such as an IUD or Depo-Provera ― prevents implantation, then no abortion takes place. Under the new definition, abortion would only occur if a chemical or device killed a preborn child who had already implanted in the endometrium (lining) of the uterus.
The pro-abortionists’ continuing agitation for a change in terminology finally bore fruit in 1965, when the American College of Obstetrics and Gynecology (ACOG) published its first Terminology Bulletin, stating, “Conception is the implantation of a fertilized ovum.” This semantic subterfuge resulted in the Bulletin inventing two misleading terms for early abortion: “Post-conceptive contraception” and “post-conceptive fertility control.”18
The deception by the medical establishment regarding the definition of “conception” coincided exactly with its devaluation of the preborn child. Neither change in attitude nor terminology was based upon some revolutionary discovery in medical technology or knowledge: the changes were made purely to further the anti-life goals of the medical profession and the pro-abortionists.
The IUD and Population Control
IUDs have confronted the anti-fertility industry with a number of forbidding problems, the greatest of which was the proven danger of the devices. This did not stop the IUD manufacturers from turning a tidy profit by dumping their products on the poor women of developing countries.19
Despite being faced with an avalanche of lawsuits claiming damage from its Copper-7 and Tatum-T IUDs, Searle said that it would continue to make IUDs for women in developing countries under population control programs funded by the United States and other Western countries.20 This is yet another example of the West’s “contraceptive imperialism” ― a willingness to dump unsafe and even lethal products on poor women of the developing world after judging them to be too risky for Western women.
Health risks associated with IUDs are a serious matter with grave implications that are not immediately evident. All of the IUDs currently on the market have patient information pamphlets that warn about the possibility of uterine perforation, especially among women with unusually-shaped uteri, or among women who have IUDs inserted immediately after childbearing. This shows that IUDs are especially ill-suited for use in developing countries, where anemia and malnutrition are often endemic, especially among women and children.21
Naturally, long-acting birth control that women cannot control is ideal for use by coercive governments. For three decades, right up to the alleged end of its dreaded “one-child” program, nearly half of all Chinese women of childbearing age had IUDs implanted after their first birth because the only alternative the government gave them was immediate sterilization.22
Final Thoughts
The IUD is the perfect example of nature’s lack of tolerance for abuse. The very idea of having a piece of plastic or metal inserted into the body in order to cripple a healthy reproductive system should be repugnant to women, but the anti-life mentality has advanced so far that millions of women are willing to risk the consequences.
+ Endnotes
[1] Robert A. Hatcher, M.D., M.P.H. Contraceptive Technology [New York City, Ardent Media] (20th Edition, 2011). Chapter 7, “Intrauterine Contraceptives (IUCs), pages 147 to 192.
To look up the patient information pamphlets (PIPs) for all intrauterine devices (or for any drug, for that matter), visit the National Institute for Health’s DailyMed archives. The latest PIPs for the currently manufactured brands of IUD are Kyleena (July 2021), Liletta (April 2020), Mirena (August 2021), Paragard T 380A (September 2019), and Skyla (July 2021). All of these patient information pamphlets confirm that IUDs sometimes act by altering the endometrium, thus making implantation either impossible or much more difficult. This is an abortifacient effect.
[2] American Medical Association Committee on Human Reproduction. “Evaluation of Intrauterine Contraceptive Devices,” Journal of the American Medical Association, February 27, 1967, page 155.
[3] United States Food and Drug Administration. “Text of Required Patient Information for IUDs.” Federal Register, May 10, 1977.
[4] Kelly Cleland, Haoping Zhu, Norman Goldstuck, Linan Cheng and James Trussell. “The Efficacy of Intrauterine Devices for Emergency Contraception: A Systematic Review of 35 Years of Experience.” Human Reproduction, May 8, 2012.
[5] Norman E. Hines, Ph.D. Practical Birth-Control Methods [New York City: Viking Press, 1938], page 138.
[6] Norman E. Hines, Ph.D. Practical Birth-Control Methods [New York City: Viking Press, 1938], page 138.
[7] Rainey Horwitz. “The Dalkon Shield.” The Embryo Project Encyclopedia, January 1, 2018.
[8] E.B. Connell. “The Uterine Therapeutic System: A New Approach to Female Contraception.” Contemporary OB/GYN, June 1975, pages 49 to 55.
[9] H.J. Tatum. “The New Contraceptive: Copper Bearing IUDs.” Contemporary Obstetrics and Gynecology, January 1973, pages 61 to 63.
[10] Robert A. Hatcher, M.D., M.P.H. Contraceptive Technology [New York City, Ardent Media] (20th Edition, 2011). Chapter 7, “Intrauterine Contraceptives (IUCs), pages 147 to 192.
[11] Harold Dubrow, M.D., and Alan Guttmacher, M.D. “The Present Status of Contraception.” Mt. Sinai Journal, 26:118-124. 1959.
[12] “The Dalkon Shield and the Questions of Safety.” Medical World News, September 13, 1974, pages 58 to 61. Also see Catherine Breslin. “Day of Reckoning.” Ms. Magazine, June 1989, pages 46 to 52. This article describes the inside story of the Dalkon Shield disaster.
[13] H.S. Kahn and C.W. Tyler. “IUD-Related Hospitalizations: United States and Puerto Rico, 1973.” Journal of the American Medical Association. 234:53-56(1973).
[14] “American Public Health Association Launches Campaign to Save IUD.” ALL News, March 9, 1987.
[15] S.D. Mumford and E. Kessel. “Was the Dalkon Shield a Safe and Effective Intrauterine Device? The Conflict between Case-Control and Clinical Trial Study Findings.” Fertility and Sterility, June 1992, pages 1,151 to 1,176.
[16] Kimberly Daniels, Ph.D., and Joyce C. Abma, Ph.D. “Current Contraceptive Status Among Women Aged 15–49: United States, 2015–2017.” National Center for Health Statistics, Centers for Disease Control, Data Brief Number 327 [December 2018]. Data table for Figure 2, “Percent Distribution of Women Aged 15–49, by Current Contraceptive Status: United States, 2015–2017.”
[17] Public Health Service leaflet No. 1066, United States Department of Health, Education and Welfare [HEW], 1963, page 27.
[18] American College of Obstetrics and Gynecology (ACOG). Terminology Bulletin, “Terms Used in Reference to the Fetus” [Chicago: ACOG], September 1965.
[19] Filipino women have probably suffered more abuse from American and European pharmaceutical companies than the women of any other nation, and this is recognized in the Filipino press. For example, one article stated that, “Many drugs and devices which are not even permitted in the United States have found their way to Manila where they are dumped on hapless Filipino women. One example was the IUD (or intrauterine device) which is associated with a great deal of disease and despair” [Max Ricketts, Mabuhay Times, April 16-29, 1991, page 12].
The Philippines is one of several nations where I have personal experience with first-world “contraceptive imperialism.” When I visited Davao City in 1995, the population suppression effort was in full swing. Propaganda posters promoting “family planning” were painted on schoolyard walls, usually prominently featuring a Catholic priest, to give the false impression that the Church supports contraception. On October 22, I visited a little shed where the Dominican sisters near Davao City perform medical procedures. These religious sisters, who are also qualified as medical doctors, pulled intrauterine devices out of women all day every Tuesday. In the late 1980s, an international population control group conducted a “Safe Motherhood Initiative” in the rural areas surrounding Davao City. They offered to give free pelvic examinations to all women between the ages of 15 to 44. While doing so, they secretly inserted IUDs into all of them! Some villages in this area therefore have no children at all under the age of about seven. As the sign on the shed door indicated, the sisters pull IUDs out of women for 25 pesos (about one dollar). If they do not have the money, they do not have to pay. Often the sister/doctor must work for a long time to remove them because the Dalkon Shields or other IUDs have embedded themselves deeply in the wall of the uterus.
[20] Front Line Updates. “Searle Removes IUDs from U.S. Market.” NRL News, February 27, 1986, page 4.
[21] Uterine perforation is one of the complications listed in the patient information pamphlets for Kyleena (July 2021); Liletta (April 2020); Mirena (August 2021); Paragard T 380A (September 2019); and Skyla (July 2021).
[22] Sui-Lee Wee. “After One-Child Policy, Outrage at China’s Offer to Remove IUDs.” The New York Times, January 7, 2017.
Related Content
Dr. Brian Clowes has been HLI’s director of research since 1995 and is one of the most accomplished and respected intellectuals in the international pro-life movement. Best known as author of the most exhaustive pro-life informational resource volume The Facts of Life, and for his Pro-Life Basic Training Course, Brian is the author of nine books and over 500 scholarly and popular articles, and has traveled to 70 countries on six continents as a pro-life speaker, educator and trainer.







Pro-life ob/gyn here. Sadly there is a lot of misinformation in this article, and information taken out of context. The authors state that no new technology has emerged since IUDs were introduced and this is simply not true. The new Miudella copper IUD was released in 2025, available in Canada and Europe and was approved by the FDA in 2025. It has half the copper of the Paragard IUD, which will reduce inflammation in the uterus and side effects of heavy bleeding. Additionally, they state that the mechanism of action of copper IUD is thinning of the lining which happens after a menstrual period, just like women who do not have an IUD. However the way a copper IUD works is disabling sperm. It does NOT prevent ovulation. The copper IUD disables sperm by causing a reaction to the head of the sperm making it unable to fertilize an egg. It does cause an inflammatory response inside the uterus, so that if a sperm is NOT disabled, it is true that a fertilized egg may have difficulty implanting. the risk of this happening is approximately 0.8%. The progesterone IUD (Mirena, Liletta, Skyla, Kylena) work by thickening cervical mucus and not letting sperm in at all. Additionally it prevents ovulation, and this is how it works as emergency contraception- by delaying ovulation, but only if inserted in the first 5 days after intercourse. It is not recommended to be inserted for EC 2 weeks after intercourse like the authors state. If a sperm makes it past the cervical mucus, and somehow a woman ovulates, the IUD also slows down the egg traveling through the fallopian tube, so less likely to be fertilized. It is associated with a higher risk of ectopic pregnancy, but even this is still lower than women not using any hormonal contraception. The pregnancy rate is 0.2% for Mirena IUD.
Thank you so much for this information about the IUD. For many years, many years ago, my husband and I were certified
teachers for Couple to Couple League, and at every natural family planning class (sympto-thermal method) we had a brochure about the
Pill and IUD. Sadly, no longer available. Recently wrote to CCL asking about an update about the IUD and they referred us
to you. So very grateful!
Have an acquaintance (a hospital worker) who claims that the IUD is not abortifacient. Again, thank you for this updated information. Sadly, those who manufacture/promote such an unnatural method of family planning will have to face God (our Creator) someday. Sincerely in Christ, Ann Marie Johnston (Mrs. Gerald)
Wow I didn’t know this. This is a big shock. I’ve heard of IUDs being used as a birth control method but I’ve never thought of or heard that it prevents the implantation of life after conception. I was considering an IUD as a method to prevent conception/pregnancy, but now that I know it is an abortifacient I would never consider it. Thank you for this vital and critical information!
Is the e-mail address you give HLI.HLI.org correct? When I attempted to send to this address it would not take.
Hi Tim, There must be a typo or other issue somewhere. HLI’s email address is hli@hli.org (HLI at HLI dot org).
I have read that recent studies since Brian Clowes’ “Abortifacient Brief: The Intrauterine Device” was written in 2017 indicate that the IUD works mostly by preventing fertilization and rarely prevents implantation. I have told many people since 2017 that the IUD is abortifacient. What should I say now? Any information would be appreciated!
Hello Tim, sadly, we have learned that politics have even seeped into medical articles, especially since the American Medical Association has taken a hardline regarding supporting abortion under any circumstances. This means that it can be very revealing to look at the affiliations of any author of any medical article that impinges upon “reproductive rights” or homosexuality/transgenderism in any way. Most such articles are written by people who have an obvious interest in such matters, and should be viewed with an extreme degree of suspicion.
There can be no doubt that IUDs are abortifacient in one of their three modes of action. If you go to the National Institute for Health’s DailyMed Web site: http://dailymed.nlm.nih.gov/dailymed/, there is a huge database of Patient Information Pamphlets (PIPs) for virtually every drug and medical device sold in the United States. These PIPs are written by the manufacturers and pharmaceutical corporations themselves. Regarding IUDs, there can be no quibbling about their modes of action, since the makers themselves use language like “alteration of the endometrium” and “prevention of implantation.” There are many types of IUDs, so I just looked at the latest available PIPs from three of the most popular: ParaGard (copper) PIP of September 2019, Mirena of August 2021 and Skyla of July 2021. All show that their IUDs are abortifacient at least part of the time, although it is impossible to know how often early abortions happen. Look under Paragraph 12.1, “Clinical Pharmacology/Mechanism of Action” for each one.
Hola, el simple hecho de que pueda ocasionar un aborto aunque sea en cada mil usuarias del dispositivo intrauterino, hace que sea un método épicamente reprobable